READ THE FULL PLX RESEARCH REPORT
Topline Results from BALANCE
Protalix Biotherapeutics, Inc. PLX began the second quarter with good news from its BALANCE trial evaluating PRX-102 against Fabrazyme. In an April 4th press release, Protalix and partner Chiesi Global Rare Diseases announced topline data from the study. The BALANCE trial met its primary endpoint and the drug showed a favorable tolerability and immunogenicity profile. Data from the successful study will be included in the planned biologics license application (BLA) submission to the FDA expected in 2H:22.
The 24-month study evaluated renal function in Fabry patients with progressing kidney disease. It enrolled 78 patients and was designed to evaluate the safety and efficacy of 1 mg/kg of PRX–102 (pegunigalsidase alfa) administered every two weeks compared to Fabrazyme (agalsidase beta).
Topline results demonstrated non-inferiority of the primary endpoint of kidney function in a head–to–head active comparison on both the Intent–to–Treat (ITT) and Per Protocol (PP) analysis sets. The median estimated glomerular filtration rate (eGFR) slope for PRX-102 was -2.514 mL/min/1.73 m2/year compared to Fabrazyme at -2.155. Th›e -0.359 difference presented an associated confidence interval of -2.444 and 1.726 and demonstrates non-inferiority of PRX-102 compared to Fabrazyme.
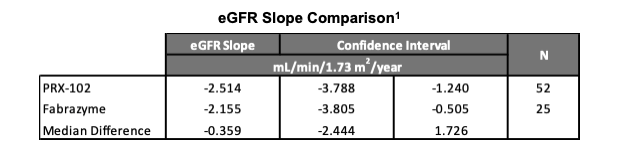
Results from the BALANCE trial will be submitted as part of the resubmission of a BLA to the FDA. Last autumn, as part of a Type A End–of–Review meeting, the FDA agreed that successful BALANCE results demonstrating non-inferiority to agalsidase beta have the potential to support the approval of PRX–102 for the treatment of Fabry disease. Results from the BALANCE trial will also be submitted to the EMA during the clock stop question period following day 120 of its review.
Safety Profile of BALANCE
Forty-seven (90.4%) patients in the PRX-102 arm experienced at least one adverse event vs. twenty-four (96.0%) using Fabrazyme. Per 100 years of exposure, this equates to 572 events for PRX-102 and 817 events for Fabrazyme. Treatment-related adverse events occurred in twenty-one (40.4%) patients taking PRX-102 vs. eleven (44%) administered Fabrazyme. Per 100 years exposure, this represents 42.8 events for PRX-102 and 153 events for Fabrazyme.
With respect to anti-drug antibodies (ADA), a lower proportion of PRX-102 subjects expressed ADA compared with the Fabrazyme group and of the subjects with ADA, fewer of the PRX-102 subjects generated the neutralizing antibodies vs. Fabrazyme at the end of the study.

Use of infusion pre-medication to prevent infusion-related reactions can help reduce allergy symptoms. During the study, both groups reduced the proportion of the pre-medication during the course of the study with PRX-102 having only 6.4% of patients using it compared with Fabrazyme at 12.5%. Even with a lower rate of the use of this medicine, the PRX-102 reported fewer infusion-related reactions compared with Fabrazyme.
Seventy-eight patients were randomized in the study, and six discontinued the study. Five of them were from the PRX-102 arm, but only one of the withdrawals was attributed to a treatment-related adverse event. No deaths occurred in the trial. Patients that completed the trial were given the opportunity to continue receiving PRX-102 bi-weekly in a long term extension study. 69 of 77 patients elected to continue in the extension.
PRX-102 demonstrated non-inferior efficacy and favorable safety in the BALANCE trial, providing an improved median reduction in eGFR slope over the 24 months of the trial compared with Fabrazyme. Protalix’ product offers a new opportunity for Fabry patients that experience difficulties with other treatments. PRX-102 benefits compared with Fabrazyme include:
➢ Shorter infusion time
➢ Potential for 4 week interval of infusion vs. 2 week
➢ Improved immunogenicity
➢ Reduced severity of side effects
➢ Superior eGFR slope
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
1. Source: Protalix data compiled by Zacks analyst
2. Source: Protalix data compiled by Zacks analyst
Read More: PLX: Topline Results from BALANCE – Benzinga



































